Sex Differences
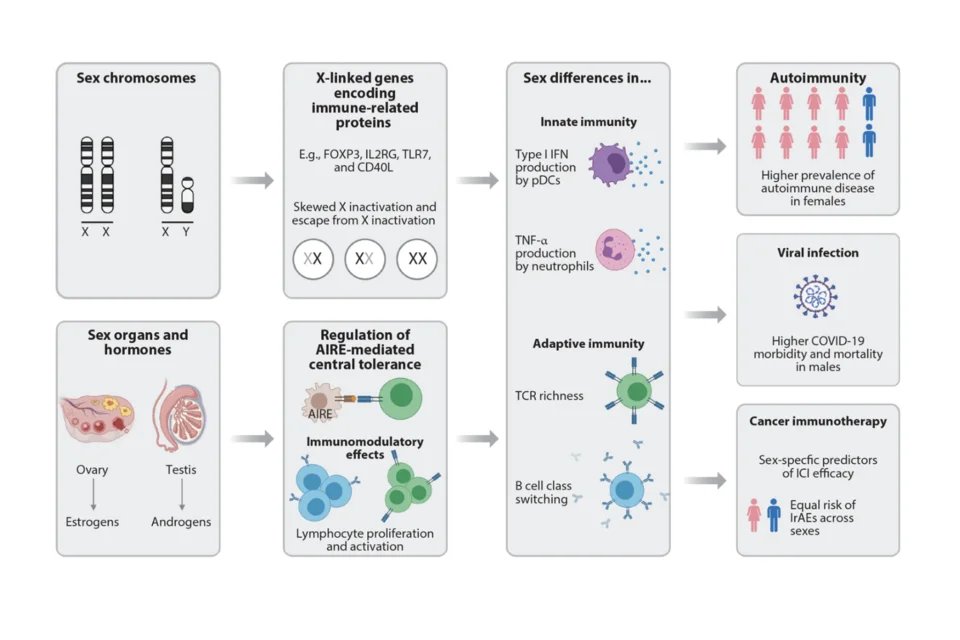
Sex Differences in Immunity
Strong epidemiological evidence now exists that sex is an important biologic variable in immunity. Recent studies, for example, have revealed that sex differences are associated with the severity of symptoms and mortality due to coronavirus disease 2019 (COVID-19). Despite this evidence, much remains to be learned about the mechanisms underlying associations between sex differences and immune-mediated conditions. A growing body of experimental data has made significant inroads into understanding sex-influenced immune responses. As physicians seek to provide more targeted patient care, it is critical to understand how sex-defining factors (e.g., chromosomes, gonadal hormones) alter immune responses in health and disease. In this review, we highlight recent insights into sex differences in autoimmunity; virus infection, specifically severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection; and cancer immunotherapy. A deeper understanding of underlying mechanisms will allow the development of a sex-based approach to disease screening and treatment. Read the review: Wilkinson et al, Annual Review of Immunology, 2022
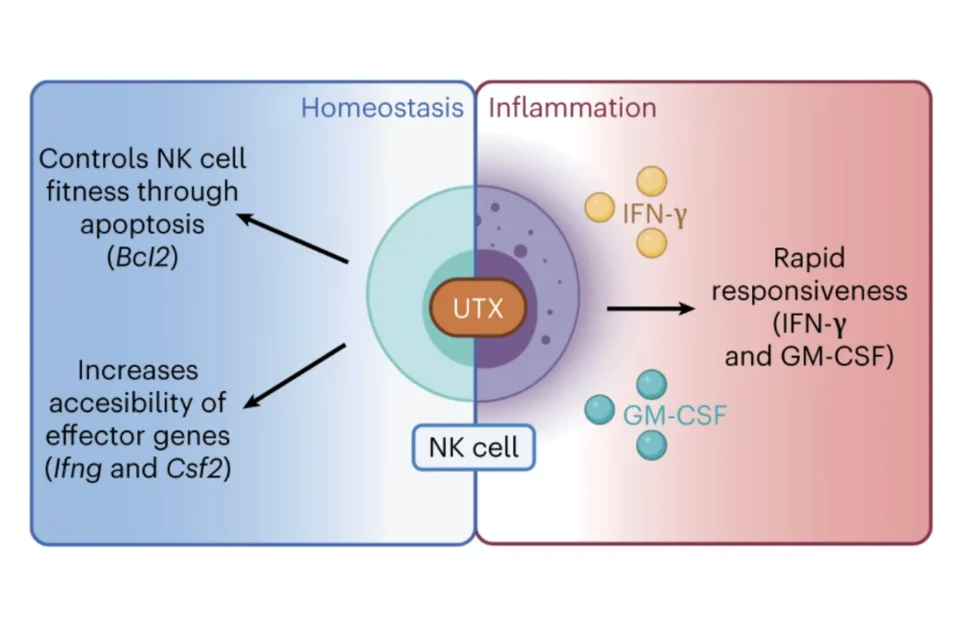
The X-linked epigenetic regulator UTX controls NK cell-intrinsic sex differences
Viral infection outcomes are sex biased, with males generally more susceptible than females. Paradoxically, the numbers of antiviral natural killer (NK) cells are increased in males. We demonstrated that while numbers of NK cells are increased in male mice, they display decreased effector function compared to females in mice and humans. These differences were not solely dependent on gonadal hormones, because they persisted in gonadectomized mice. Kdm6a (which encodes the protein UTX), an epigenetic regulator that escapes X inactivation, was lower in male NK cells, while NK cell-intrinsic UTX deficiency in female mice increased NK cell numbers and reduced effector responses. Furthermore, mice with NK cell-intrinsic UTX deficiency showed increased lethality to mouse cytomegalovirus. Integrative multi-omics analysis revealed a critical role for UTX in regulating chromatin accessibility and gene expression critical for NK cell homeostasis and effector function. Collectively, these data implicate UTX as a critical molecular determinant of sex differences in NK cells. Read the paper: Cheng et al, Nature Immunology, 2023
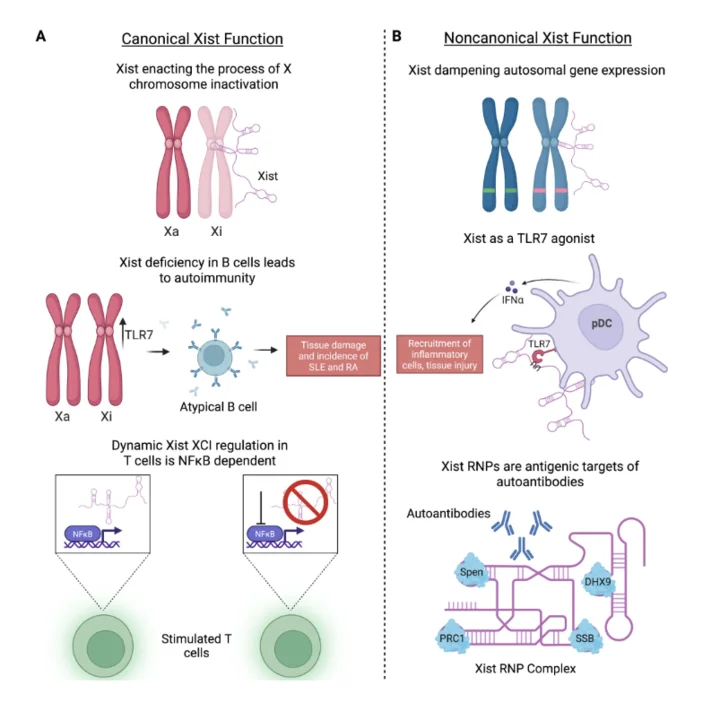
Sex Matters: Hormonal and Chromosomal Determinants
of Autoimmunity and Anti-Cancer Immunity Across
the Lifespan
Sex plays a key role in shaping both anti‐cancer immunity and autoimmunity. Biological factors underlying sexual dimorphism have now been identified in multiple aspects of anti‐cancer immunity and autoimmunity. These factors include sex differences in hormone levels, chromosome complement, and expression of the long non‐coding RNA XIST. In this review, we discuss recent advances delineating how these differences alter immune responses against cancer and autoimmune responses against healthy tissues. Moreover, we now understand that hormone levels change (e.g., in mini‐puberty, menopause, and andropause) and that somatic alterations in chromosomal complement accumulate (e.g., loss of Y [LOY] chromosome) across the lifespan. We also include here a discussion of how these changes affect anti‐cancer immunity and autoimmunity across a lifetime. These recent advances will set the stage for identifying immunotherapeutic approaches that optimize anti‐cancer immunity while controlling the autoimmune responses. Read our invited review: Bustillos et al, Immunological Reviews, 2026